Usually when we talk about colors, we talk about pigments, which are giving objects their appearance. But there are other ways to generate colorful images. In nature we can find multiple examples for structure related colors, most commonly known are the butterflies and peacocks. This week I try to give an answer to the question: “ What is the relation between colors and structures?“
Pigments
First I want to start with pigments, just to clarify the differences between them and structure related images.
Melanin is well known as the pigment giving our skin its color. It is located in our cells and fulfils an important task: protecting cells from UV radiation. [1] Another example, which will show that pigments can have other purposes than just giving color is chlorophyll.
No further explanations about its tasks are needed, because we all had discussed this topic in school. Do you remember? It was the time where all answers, your biology teacher wanted to hear, included „photosynthesis!“[2]
In general, pigments are inside cells, whereas structures are on the surface generating a color.
Again, I left many things open for you, but at this point I find it more interesting to talk about the chlorophyll’s behavior to appear green.
Light Absorption
I sit here in front of my notebook and imagine a tree in our garden at home, in the morning of a summer day. This powerful green feels really good. But then my mind switches to the one and only question: Why? Okay, here is the cold truth. Chlorophyll in general has two wavelength peaks with good absorption rates. The first is around 600 to 800 nm and the second is around 400 to 500 nm.
- by Aushulz derivative work: M0tty [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0) or GFDL (http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons
Now you can imagine what happens to my tree with its leaves in the garden. The sunlight with its full color spectrum hits the leaves. Blue color spectrum gets absorbed around 400 nm and the red one around 700 nm. I am sitting here in front of my display and have only just three letters in my mind: R G B, Red, Green and Blue.
Red and Blue are absorbed, leaving green to reflect. (It is called green gap, you can figured it out by look at the picture) [3]
And that’s it. We see green leaves.
Light and Structures
A combination of absorption and reflection generates a certain image in a specific color, is what happens to the leaf. But, is it possible to generate a color without absorption of specific color spectra? Oh yes, that’s quite possible. The keyword for that could be – photonic crystal. It is a periodic optical nanostructure. They exist in nature and are more and more used in modern technology. When I think of an appearance in nature, I think of opals, shells (nacre, we discussed it in one of Anja’s articles) or butterflies.
For technology I have to think about waveguides or optical fibers and sensors. The clue is the nanostructure which can direct, fracture or reflect specific wavelengths of light spectrum.
Structures and Colors
Different structure can lead to different results in terms of appearance. It can be a very intense color or an interference color, depending on the point of view. You can filter light with structures or you capture it. That way, an object will stay black, because no light escapes out of its structure.
I saw two parabolic mirrors with a setup, where light can go in the system but never leaves it. The light is endlessly reflected and finally absorbed by air. When you are watching the system from outside, it is the deepest black you can think of.
Here is another simple example of light capturing.

- by borda at www.deviantart.com (Black Hole’s Magic Mirror)
Okay, these examples were not caused by a nanostructure, but they were good to show the underlying principle. Nanostructure are also not hard to imagine.
You already heard about the absorption of specific light wavelengths by pigments. A structure could do the same but it is not absorbing, it is fracturing light. This can lead to a similar effects, because fractured light and reflected light have different velocities inside specific media.
An example:
In a dust cloud, longer wavelengths get much less disrupted than the short wavelengths. This leads to a yellowish color of such a cloud. In scattered light, the dust cloud will appear more bluish.
Without going to much in technical details of all this, I try to show you how we can use this to create useful sensor devices.
Sensors using nanostructures
In this part I will explain the general concept of a gas concentration sensor using photonic crystals [4]. You need a source of light and a nanostructure generating a reflection of light in a specific wavelength. This specific wavelength depends on the gas that you want to detect, because the molecules of a certain gas can absorb or at least fracture light. It also depends on another rule, known as Kirchhoff’s law of thermal radiation (a good absorber is a good emitter, etc.). Carbon dioxide, for example, is capable to absorb infrared light (wavelengths between 780 nm and 1000 nm).
General considerations are set and now you can see in the next picture how a possible measurement setup could look like. The light source brings light into the system and will be detected by a light detector. If the concentration of a specific gas is high, then the detected light intensity is low.
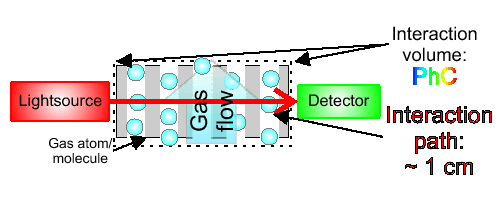
- by http://www-old.mpi-halle.mpg.de/ (Max-Planck-Institut – microstructure physics)
At a normal scale the light source is usually a infrared bulb or something similar. But if you want to decrease the size of a system like this, you could use photonic crystals to achieve a very compact sensor (maybe as small as your finger tip!?)
I hope you got the idea. For more information and details you can check the references. Especially the sources of the Wikipedia articles that I linked are very interesting. The articles itself is just an introduction.
Bye,
Jan
References:
[1] The Protective Role of Melanin Against UV Damage in Human Skin – by Michaela Brenner and Vincent J. Hearing
[2] https://en.wikipedia.org/wiki/Photosynthesis
[3] Absorption of light by chlorophyll solutions – G. Mackinney (1941)
[4] https://en.wikipedia.org/wiki/Photonic_crystal
[5] peacock feather



